Chimeric-antigen-receptor (CAR) natural-killer (CAR-NK) cells are a promising off-the-shelf alternative to CAR-T therapy. High or repeated doses are usually needed to maintain clinical efficacy, so a manufacturing platform that can deliver billions of cytolytic NK cells at low cost is critical. On 7 October 2025, Jinyong Wang, Mengyun Zhang (Institute of Zoology, Chinese Academy of Sciences) and Xiaofan Zhu (Institute of Hematology & Blood Diseases Hospital, CAMS) published a paper in Nature Biomedical Engineering (impact factor 26.6) entitled “Large-scale generation of iNK and CAR-iNK cells from CD34⁺ haematopoietic stem and progenitor cells for adoptive immunotherapy.” They describe a three-step protocol that starts from human umbilical-cord-blood CD34⁺ haematopoietic stem and progenitor cells (CB-CD34⁺ HSPCs) and yields clinical-scale lots of induced NK (iNK) or CAR-iNK cells. A single cord-blood unit produced 140–830 million mature iNK cells or 70–320 million CAR-iNK cells that expressed high levels of CD16 and the CAR and contained no detectable T-cell contamination. Both fresh and thawed iNK and CAR-iNK cells killed a broad panel of human tumour lines and prolonged survival in several human tumour xenograft models. The high cell yield and the very low reagent cost support widespread adoption of this approach for cancer immunotherapy.
Cellular immunotherapy, especially CAR-T-cell therapy, has transformed the treatment of B-cell malignancies, but its high price and limited cell supply have driven interest in natural-killer cells as a universal alternative. NK cells recognise tumours through a balance of activating and inhibitory receptors and can exert direct cytotoxicity without prior sensitisation. CAR-NK cells therefore promise both antigen-specific and innate cytotoxicity with reduced risk of graft-versus-host or cytokine-release toxicities.
Human NK cells circulate with a half-life of roughly two weeks, and allogeneic NK products typically persist for only a few days to a few weeks in patients (median ≈ 7 days). Clinical protocols therefore call for high (≥ 10⁷ cells kg⁻¹) and often repeated doses, necessitating manufacturing platforms that can generate very large cell numbers at acceptable cost.
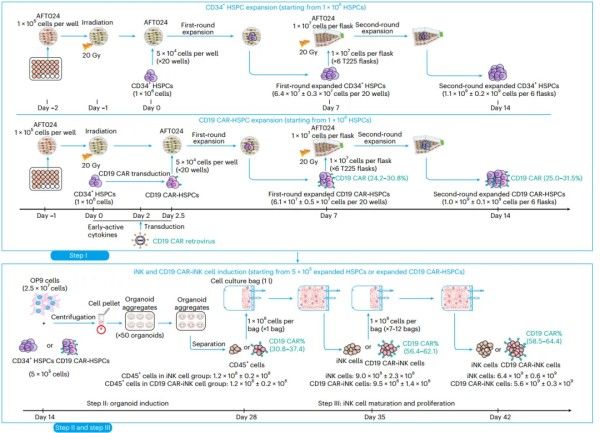
CD34⁺ HSPCs can differentiate into all blood lineages, including NK cells. Cytokine-based culture systems have been reported to yield 1,879–4,450 NK cells per input HSPC, but the resulting cells express little CD16 (≈ 3 %), limiting antibody-dependent cellular cytotoxicity (ADCC). Engineering HSPCs with a CAR before lymphoid differentiation has also been explored, and recent organoid-aggregate cultures that combine stem cells with feeder cells can markedly boost differentiation efficiency. The authors therefore set out to integrate high-density HSPC expansion with organoid-driven lymphoid specification and subsequent NK maturation in a single closed-system pipeline.
Starting from either naive CB-CD34⁺ HSPCs or the same cells transduced with a CD19-CAR retroviral vector, the protocol proceeds through three stages: (1) expansion of CD34⁺ cells for 14 days in cytokine-rich serum-free medium; (2) lineage differentiation within feeder-free organoid aggregates from day 14 to 28; and (3) maturation and large-scale proliferation in gas-permeable culture bags from day 28 to 49. By day 42, a single input CD34⁺ cell had given rise to 1.4 × 10⁷ ± 0.1 × 10⁷ iNK cells or 7.6 × 10⁶ ± 1.2 × 10⁶ CD19-CAR-iNK cells; final harvest on day 49 yielded up to 8.3 × 10⁷ ± 0.7 × 10⁷ iNK cells or 3.2 × 10⁷ ± 0.2 × 10⁷ CAR-iNK cells. Extrapolating to the 1–2 × 10⁶ CD34⁺ cells typically found in a cord-blood unit, the platform could produce trillions of CAR-iNK cells—enough for hundreds of patients.
Remarkably, introducing the CAR at the HSPC stage cut retrovirus consumption by a factor of 140,000–600,000 compared with conventional transduction of mature NK cells. Cryopreserved iNK and CAR-iNK cells retained full cytotoxic activity after thaw and continued to eradicate solid-tumour xenografts for up to six months in vivo.
The authors conclude that the pipeline offers a translational path to affordable, off-the-shelf CAR-iNK therapy for broad patient populations.